Testing Soft and Hard Homogenization Protocol
Soft and Hard Homogenization Protocol Testing Round 1
20190606 E.S.
Testing the soft and hard homogenization protocol on extra molecular samples from the recovery time period of the Holobiont Integration project. 2 M. capitata and 2 P. acuta fragments were randomly chosen.
Soft and Hard homogenization, and DNA/RNA Extractions followed this protocol. General Zymo Duet RNA DNA Extraction protocol found here.
Original samples:
| ORIGIN | Site.Name | POINT | SITE.ID | Species | COLLECT.DATE | PLUG.ID | TANK# | TREATMENT | ANALYSIS | TIME | TIME POINT | SAMPLING.DATE | Dead.Alive | NOTES | Sample Location | Shipment Date | Lab Work | Lab Work by | Lab Work Date |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SITE 4 | Reef.11.13 | 6 | P60 | Pacuta | 20180907 | 1431 | 6 | ATAC | Molecular | 11:24 | 11 | 20181215 | Alive | In Tank 6 20181118; off plug | In Transit to URI | 20190206 | Soft and hard homogenization | ES | 20190606 |
| SITE 1 | Lilipuna.Fringe | 6 | M54 | Mcapitata | 20180908 | 1591 | 8 | ATAC | Molecular | 10:12 | 11 | 20181215 | Alive | In Tank 8 20181118 | In Transit to URI | 20190206 | Soft and hard homogenization | ES | 20190606 |
| SITE 3 | Reef.18 | 5 | M45 | Mcapitata | 20180910 | 2309 | 6 | ATAC | Molecular | 11:28 | 11 | 20181215 | Alive | In Tank 6 20181118; off plug | In Transit to URI | 20190206 | Soft and hard homogenization | ES | 20190606 |
| SITE 3 | Reef.18 | 8 | P72 | Pacuta | 20180910 | 2364 | 6 | ATAC | Molecular | 11:27 | 11 | 20181215 | Alive | In Tank 6 20181118 | In Transit to URI | 20190206 | Soft and hard homogenization | ES | 20190606 |
Observations:
- Vortexing for two minutes was more than enough time. The P. acuta tissue came off quicker than M. capitata. This might cause problems later in the protocol. Maybe less beads or more fragment pieces?
- Both homogenization steps got at least 500 uL of sample.
- Almost all of the tissue from fragment 1431 came off during the “soft” homogenization step.
- After hard homogenization, not all of the tissue from the M. capitata came off the skeleton, but the liquid was dark brown.
- Tissue Lyser was broken, so bead bug was used for the “hard” homogenization step instead. Run at 400 speed and 30 seconds.
- Heating started at 12:55, ended at 2:25.
Changes to protocol:
- Half of the beads from bead bug machine tubes were used.
- 500 uL of shield used instead of 1000 uL for the “hard” homogenization step.
- 30 uL of Proteinase K Buffer, and 15 uL of Proteinase K used.
- Proteinase K buffer and Proteinase K added after bead beating steps.
Calculations:
DNAse I
- 75 uL buffer x 8 samples = 600 uL buffer
- 5 uL DNAse I x 8 samples = 40 uL DNAse I
Qubit Master Mix (x2 for DNA and RNA)
- 8 samples + 2 standards + 0.2% error = 10.2 uL of Quant-IT Reagent
- 199 x 10.2 = 2,029.8 uL buffer
2 standards: 190 uL Master Mix + 10 uL standard
16 samples: 199 uL Master Mix + 1 uL sample
15 minute incubation started at 15:44
- Re-aliquoted fragment 1591 hard and soft DNA and RNA tubes
Qubit (Protocol)Results
dsDNA broad range
| DNA(ng/uL) | Trial 1 | Trial 2 | Avg |
|---|---|---|---|
| Standard 1 | 174.13 | NA | NA |
| Standard 2 | 18,408.70 | NA | NA |
| 1591 Hard | 109 | 107 | 108 |
| 1591 Soft | 54.4 | 53.8 | 54.1 |
| 1431 Hard | 130 | 130 | 130 |
| 1431 Soft | 193 | 192 | 192.5 |
| 2309 Hard | 65.8 | 65.2 | 65.5 |
| 2309 Soft | 47.6 | 47.2 | 47.4 |
| 2364 Hard | 199 | 198 | 198.5 |
| 2364 Soft | 55.4 | 54.8 | 55.1 |
RNA broad range
| RNA (ng/uL) | Trial 1 | Trial 2 | Avg |
|---|---|---|---|
| Standard 1 | 396.31 | NA | NA |
| Standard 2 | 10,506.90 | NA | NA |
| 1591 Hard | 66.2 | 66.2 | 66.2 |
| 1591 Soft | 73.2 | 73 | 73.1 |
| 1431 Hard | 158 | 158 | 158 |
| 1431 Soft | 274 | 274 | 274 |
| 2309 Hard | 75.6 | 75.4 | 75.5 |
| 2309 Soft | 32.4 | 32.4 | 32.4 |
| 2364 Hard | 193 | 193 | 193 |
| 2364 Soft | 202 | 202 | 202 |
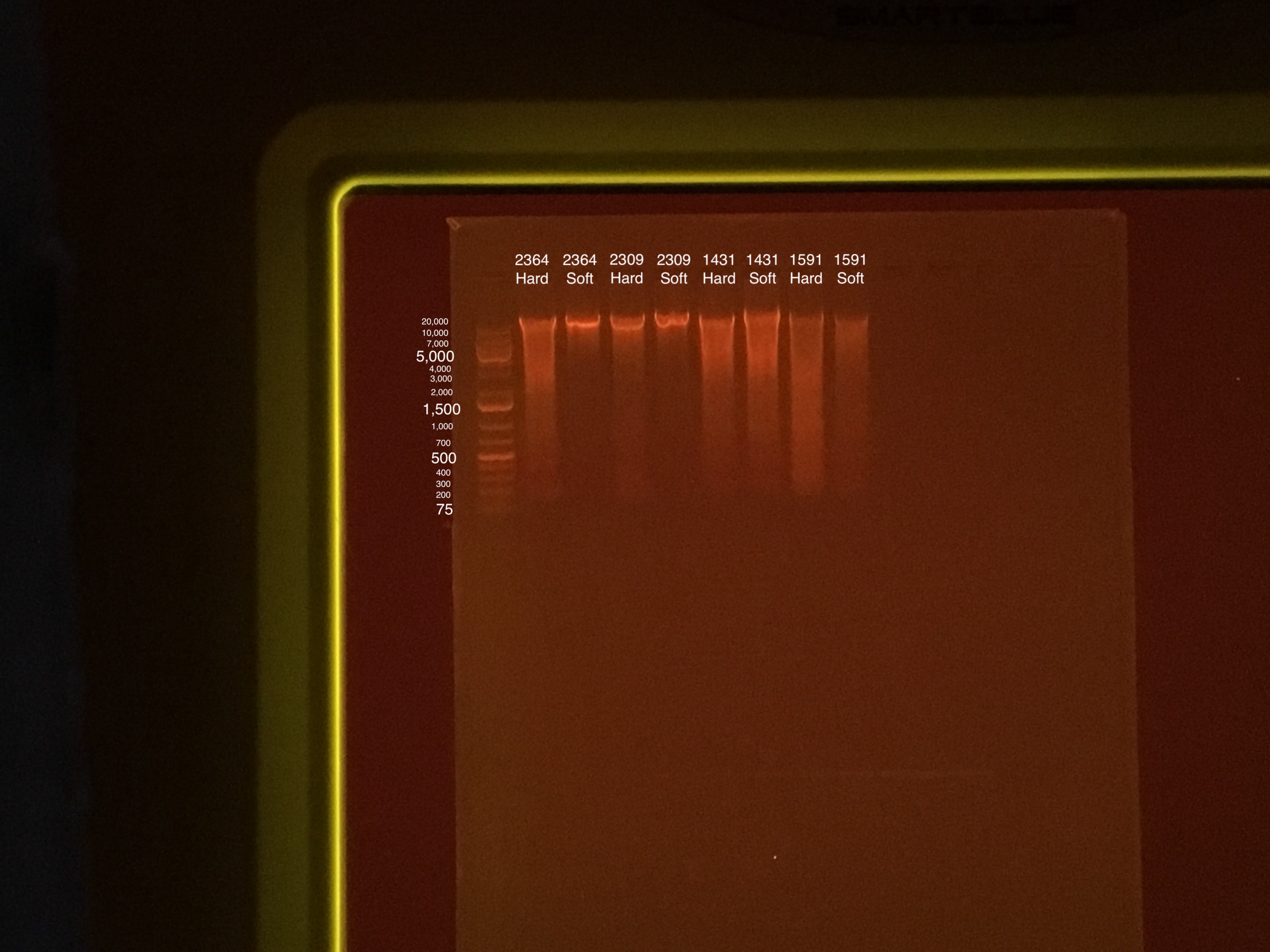
Gel Electrophoresis (Protocol):
- Gel made during 15 minute incubation period at 15:58
- 100V for 45 minutes
- 75 uL TAE buffer + 0.75 g agarose + 5 uL of Biotium gel RED gel stain to make a 1.5% gel
- 5 uL samples + 1 uL 6x purple loading dye
- 4 uL GeneRuler 1 kb Plus DNA ladder + 1 uL 6x purple loading dye
Sample order: Ladder, 2364 Hard, 2364 Soft, 2309 Hard, 2309 Soft, 1431 Hard, 1431 Soft, 1591 Hard, 1591 Soft
TapeStation (Protocol) Results:
- Thermocycler (rna denature program): 3 minutes at 72 °C, 2 minutes at 4 °C, hold at 4 °C. start 18:15 end 18:21
- TapeStation start 18:28 end 18:38
- Ladder: Agilen RNA screentape ladder
| Coral ID | Tape Station ID | RIN^e |
|---|---|---|
| Ladder | A1 | NA |
| 1591 Hard | B1 | 3.3 |
| 1591 Soft | C1 | 4 |
| 1431 Hard | D1 | 7 |
| 1431 Soft | E1 | 7.7 |
| 2309 Hard | F1 | 7.1 |
| 2309 Soft | G1 | 8.3 |
| 2364 Hard | H1 | 4.1 |
| 2364 Soft | A2 | 6.9 |
Link to 20190606 report
Link to Agilent 4200 TapeStation System
